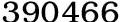Theoretical Impact: Formula: Eg=[28.8/(2(XM-XN)2)1/4*(1-f12/1+2*f12)]POWER (XM/XN)2 Where:f12=[4pN/3]*[aM12*r12]/M12 Electro Negativity values of Elemental Semiconductors: Compound Al Ga As In P Sb N E.N value 1.5 1.8 2 1.7 2.1 1.9 3
Electro Negativity values of GaAsxN1-x III-V Ternary Semiconductor
X value 0 0.1 0.15 0.2 0.25 0.3 0.35 0.4 0.45 0.5 1-x value 1 0.9 0.85 0.8 0.75 0.7 0.65 0.6 0.55 0.5 Compound GaAsxN1-x XM value 1.9 1.9 1.9 1.9 1.9 1.9 1.9 1.9 1.9 1.9 XN value 3 2.880794 2.822978 2.76632 2.710806 2.656402 2.603091 2.550849 2.499655669 2.44949 (XM/XN)2 0.4011111 0.434994 0.452994 0.47174 0.4912588 0.511587 0.532756 0.554802 0.577759141 0.601667 (XM-XN)2 1.21 0.961956 0.851889 0.75052 0.6574064 0.572145 0.494337 0.423604 0.359586921 0.301939 2(XM-XN)2 2.3133764 1.947949 1.804863 1.6824 1.5772446 1.486732 1.408673 1.341274 1.283058474 1.2328 (2(XM-XN)2)1/4 1.2332797 1.181393 1.159074 1.13889 1.1206623 1.104227 1.089438 1.076166 1.064293996 1.053715 28.8/(2(XM-XN)2)1/4 23.352367 24.378 24.84743 25.2878 25.699089 26.0816 26.43565 26.76166 27.06019211 27.33187
ALPHA-M 37.49 42.016 44.279 46.542 48.805 51.068 53.331 55.594 57.857 60.12 RO-VALUES 6.1 6.021 5.9815 5.942 5.9025 5.863 5.8235 5.784 5.7445 5.705 M-VALUES 83.73 89.821 92.8665 95.912 98.9575 102.003 105.0485 108.094 111.1395 114.185 ALPHA-M*RO/M 2.7312672 2.816472 2.851995 2.8834 2.911063 2.935322 2.956473 2.974778 2.990471763 3.003762
TOTAL 4*PI*N 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 4*PI*N/3 VALUES 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 (4PIN/3)*ALPHAM*RO/M 6.887E+24 7.1E+24 7.19E+24 7.3E+24 7.341E+24 7.4E+24 7.46E+24 7.5E+24 7.54086E+24 7.57E+24
1-(4PIN/3)*ALPHAM*RO/M 6.887E+24 7.1E+24 7.19E+24 7.3E+24 7.341E+24 7.4E+24 7.46E+24 7.5E+24 7.54086E+24 7.57E+24 1+2*(4PIN/3)*ALPHAM*RO/M 1.377E+25 1.42E+25 1.44E+25 1.5E+25 1.468E+25 1.48E+25 1.49E+25 1.5E+25 1.50817E+25 1.51E+25 1-phi12/1+phi12 0.5 0.5 0.5 0.5 0.5 0.5 0.5 0.5 0.5 0.5 28.8/(2(XM-XN)2)1/4*(1-phi12/1+2*phi12) 11.676184 12.189 12.42371 12.6439 12.849544 13.0408 13.21782 13.38083 13.53009605 13.66593
Eg value 2.6798239 2.96749 3.131042 3.30978 3.5055069 3.720276 3.956444 4.216718 4.504218239 4.822545
X value 0.55 0.6 0.65 0.7 0.75 0.8 0.85 0.9 0.95 1 1-x value 0.45 0.4 0.35 0.3 0.25 0.2 0.15 0.1 0.05 0
Compound XM value 1.9 1.9 1.9 1.9 1.9 1.9 1.9 1.9 1.9 1.9 XN value 2.400331 2.352158 2.304952 2.258694 2.213364 2.168944 2.125415 2.082759 2.04096 2
(XM/XN)2 0.626563 0.652491 0.67949 0.707608 0.736888 0.76738 0.799134 0.832202 0.866639 0.9025 (XM-XN)2 0.250331 0.204447 0.163986 0.128661 0.098197 0.072331 0.050812 0.033401 0.01987 0.01 2(XM-XN)2 1.18948 1.152245 1.120379 1.093279 1.070435 1.051414 1.035848 1.023422 1.013868 1.006956 (2(XM-XN)2)1/4 1.044334 1.036063 1.028824 1.022546 1.017162 1.012613 1.008844 1.005805 1.003449 1.001734 28.8/(2(XM-XN)2)1/4 27.57739 27.79754 27.99312 28.165 28.31408 28.44128 28.54753 28.63379 28.70101 28.75014
ALPHA-M 62.383 64.646 66.909 69.172 71.435 73.698 75.961 78.224 80.487 82.75 RO-VALUES 5.6655 5.626 5.5865 5.547 5.5075 5.468 5.4285 5.389 5.3495 5.31 M-VALUES 117.2305 120.276 123.3215 126.367 129.4125 132.458 135.5035 138.549 141.5945 144.64 ALPHA-M*RO/M 3.014837 3.023865 3.030997 3.036371 3.04011 3.042328 3.043126 3.0426 3.040833 3.037904
TOTAL 4*PI*N 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 7.56E+24 4*PI*N/3 VALUES 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 2.52E+24 (4PIN/3)*ALPHAM*RO/M 7.6E+24 7.63E+24 7.64E+24 7.66E+24 7.67E+24 7.67E+24 7.67E+24 7.67E+24 7.67E+24 7.66E+24
1-(4PIN/3)*ALPHAM*RO/M 7.6E+24 7.63E+24 7.64E+24 7.66E+24 7.67E+24 7.67E+24 7.67E+24 7.67E+24 7.67E+24 7.66E+24 1+2*(4PIN/3)*ALPHAM*RO/M 1.52E+25 1.53E+25 1.53E+25 1.53E+25 1.53E+25 1.53E+25 1.53E+25 1.53E+25 1.53E+25 1.53E+25 1-phi12/1+phi12 0.5 0.5 0.5 0.5 0.5 0.5 0.5 0.5 0.5 0.5 28.8/(2(XM-XN)2)1/4*(1-phi12/1+2*phi12) 13.7887 13.89877 13.99656 14.0825 14.15704 14.22064 14.27376 14.31689 14.3505 14.37507
Eg value 5.175873 5.569051 6.007731 6.49852 7.04917 7.668802 8.368189 9.160099 10.05972 11.08519
Doping of As component in a Binary semiconductor like GaN and changing the composition of do pant has actually resulted in lowering of Band Energy Gap.
Future Plans: 1) Current data set of Electro Negativity values of GaAsxN1-x III-V Ternary Semiconductors and Band Energy Gap values include the most recently developed methods and basis sets are continuing. The data is also being mined to reveal problems with existing theories and used to indicate where additional research needs to be done in future. 2) The technological importance of the ternary semiconductor alloy systems investigated makes an understanding of the phenomena of alloy broadening necessary, as it may be important in affecting semiconductor device performance.
Conclusion: 1) This paper needs to be addressed theoretically so that a fundamental understanding of the physics involved in such phenomenon can be obtained in spite of the importance of ternary alloys for device applications. 2) Limited theoretical work on Electro Negativity values and Band Energy Gap of GaAsxN1-x III-V Ternary Semiconductors with in the Composition range of (0 Results and Discussion: Electro Negativity values of Ternary Semiconductors are used in calculation of Band Energy Gaps and Refractive indices of Ternary Semiconductors and Band Energy Gap is used for Electrical conduction of semiconductors. This phenomenon is used in Band Gap Engineering. Acknowledgments. – This review has benefited from V.R Murthy, K.C Sathyalatha contribution who carried out the calculation of physical properties for several ternary compounds with additivity principle. It is a pleasure to acknowledge several fruitful discussions with V.R Murthy.
References: 1) IUPAC Gold Book internet edition: "Electronegativity". 2) Pauling, L. (1932). "The Nature of the Chemical Bond. IV. The Energy of Single Bonds and the Relative Electronegativity of Atoms". Journal of the American Chemical Society 54 (9): 3570–3582.. 3) Pauling, Linus (1960). Nature of the Chemical Bond. Cornell University Press. pp. 88–107. ISBN 0801403332 . 4) Greenwood, N. N.; Earnshaw, A. (1984). Chemistry of the Elements. Pergamon. p. 30. ISBN 0-08-022057-6. 5) Allred, A. L. (1961). "Electronegativity values from thermochemical data". Journal of Inorganic and Nuclear Chemistry 17 (3–4): 215–221.. 6) Mulliken, R. S. (1934). "A New Electroaffinity Scale; Together with Data on Valence States and on Valence Ionization Potentials and Electron Affinities". Journal of Chemical Physics 2: 782–793.. 7) Mulliken, R. S. (1935). "Electronic Structures of Molecules XI. Electroaffinity, Molecular Orbitals and Dipole Moments". J. Chem. Phys. 3: 573–585.. 8) Pearson, R. G. (1985). "Absolute electronegativity and absolute hardness of Lewis acids and bases". J. Am. Chem. Soc. 107: 6801.. 9) Huheey, J. E. (1978). Inorganic Chemistry (2nd Edn.). New York: Harper & Row. p. 167. 10) Allred, A. L.; Rochow, E. G. (1958). "A scale of electronegativity based on electrostatic force". Journal of Inorganic and Nuclear Chemistry 5: 264.. 11) Prasada rao., K., Hussain, O.Md., Reddy, K.T.R., Reddy, P.S., Uthana, S., Naidu, B.S. and Reddy, P.J., Optical Materials, 5, 63-68 (1996). 12) Ghosh, D.K., Samantha, L.K. and Bhar, G.C., Pramana, 23(4), 485 (1984). 13) CRC Handbook of Physics and Chemistry, 76th edition. 14) Sanderson, R. T. (1983). "Electronegativity and bond energy". Journal of the American Chemical Society 105: 2259 15) Murthy, Y.S., Naidu, B.S. and Reddy, P.J., “Material Science &Engineering,”B38, 175 (1991)
Related Articles -
Band Energy Gap, Composition, Electro Negativity, Molecular weight, density, optical polarizability,